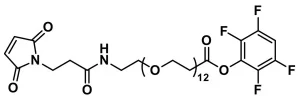
MAL-dPEG®36-NHS ester, product number QBD-10904, is a crosslinking reagent that joins a sulfhydryl to a free amine through a hydrophilic bridge. The sulfhydryl groups react with a maleimide group via a Michael addition reaction. The amines form amide bonds with the crosslinker by nucleophilic substitution of the N-hydroxysuccinimidyl (NHS) ester of a carboxylic acid group. The maleimide and NHS functional groups on the crosslinking compound sit at either end of a long (118 atoms, 134.8 Å), discrete-length polyethylene glycol (dPEG®) chain.
Reactions that join free amines with free thiols are among the most popular, most useful crosslinking reactions in bioconjugate chemistry. These reactions require heterobifunctional reagents that bridge the two groups. Traditional crosslinkers are hydrophobic molecules. Vector Laboratories’ dPEG® crosslinking products are water-soluble, amphiphilic, single molecular weight PEG compounds with discrete chain lengths.
The conjugation of conventional hydrophobic crosslinking reagents to biomolecules almost inevitably triggers problems such as aggregation and precipitation of the conjugates. These problems do not occur with our water-soluble, non-immunogenic dPEG® crosslinkers.
Because NHS esters hydrolyze readily in water or aqueous buffer, the NHS ester end of the molecule must conjugate to a target molecule before conjugating the maleimide end of the molecule. At pH 7.0 – 7.5, NHS esters react optimally with free amines. However, NHS esters can react with free amines with pH as low as 6.5. As the pH increases, the hydrolysis rate of the NHS ester increases. Indeed, at pH 8 and 25°C, the half-life of an NHS ester in aqueous media is one hour, while at pH 8.6 and 4°C, the half-life falls to ten (10) minutes.[3] Thus, we strongly discourage storing MAL-dPEG®36-NHS ester, product number QBD-10904, in water or aqueous buffer. Instead, we recommend that customers make new solutions of the product as needed, use them immediately, and discard unused solutions after use.
The reaction of the maleimide end of MAL-dPEG®36-NHS ester with a sulfhydryl proceeds optimally at pH 6.5 – 7.5. Use the lowest reasonable pH within this range. Above pH 7.5, free amines compete with free thiols at the maleimide reaction site, which can cause confusing results. Moreover, at higher pH values, the maleimide ring may open to form unreactive maleamic acid.
The use of MAL-dPEG®36-NHS ester, product number QBD-10904, has been published in numerous scientific paper and patents. The following list highlights some of the more interesting uses of this product:
development of extracellular drug conjugates (EDCs), a type of ADC for extracellular markers;
targeted delivery of cancer drugs;
as a long, flexible tether for atomic force microscopy (AFM); and
as a surface coating for amine-functionalized nanoparticle surfaces.
| Unit Size | 100mg, 1000mg |
|---|---|
| Molecular Weight | 1923.18; single compound |
| Chemical formula | C₈₆H₁₅₉N₃O₄₃ |
| CAS | N/A |
| Purity | > 95% |
| Spacers | dPEG® Spacer is 118 atoms and 134.8 Å |
| Shipping | Ambient |
| Typical solubility properties (for additional information contact Customer Support) | Methylene chloride, DMAC or DMSO or Acetonitrile. |
| Storage and handling | -20°C; Always let come to room temperature before opening; be careful to limit exposure to moisture and restore under an inert atmosphere; stock solutions can be prepared with dry solvent and kept for several days (freeze when not in use). dPEG® pegylation compounds are generally hygroscopic and should be treated as such. This will be less noticeable with liquids, but the solids will become tacky and difficult to manipulate, if care is not taken to minimize air exposure. |
Greg T. Hermanson, Bioconjugate Techniques, 2nd Edition, Elsevier Inc., Burlington, MA 01803, April, 2008 (ISBN-13: 978-0-12-370501-3; ISBN-10: 0-12-370501-0); See pp. 276-335 for general description and use of heterobifunctional crosslinkers, as well as his specific discussion with protocols of our MAL-dPEG®x-NHS esters on pp. 718-722.
Greg T. Hermanson, Bioconjugate Techniques, 3rd Edition, Elsevier, Waltham, MA 02451, 2013, ISBN 978-0-12-382239-0; See Chapter 18, Discrete PEG Reagents, pp. 787-821, for a full overview of the dPEG® products.
Applicable patents and legal notices are available at legal notices.
Stay in the Loop. Join Our Online Community
Products
Ordering
About Us
Application
Resources

©Vector Laboratories, Inc. 2025 All Rights Reserved.
To provide the best experiences, we use technologies like cookies to store and/or access device information. Consenting to these technologies will allow us to process data such as browsing behavior or unique IDs on this site. Not consenting or withdrawing consent, may adversely affect certain features and functions. Privacy Statement
How do I Request a Quote?
To request a quote for products: