CAS Number 756525-92-5 | |
Molecular Weight 839.96; single compound | |
Chemical Formula C₃₈H₆₃N₃O₁₉ | |
Purity > 98% | |
Unit Size 100 mg, 1000 mg | |
Solubility Methylene chloride, Acetonitrile, DMAC or DMSO. | |
Spacers dPEG® Spacer is 40 atoms and 46.5 Å | |
Storage Instructions -20°C; Always let come to room temperature before opening; be careful to limit exposure to moisture and restore under an inert atmosphere; stock solutions can be prepared with dry solvent and kept for several days (freeze when not in use). dPEG® pegylation compounds are generally hygroscopic and should be treated as such. This will be less noticeable with liquids, but the solids will become tacky and difficult to manipulate, if care is not taken to minimize air exposure. | |
Shipping Instructions Ambient |
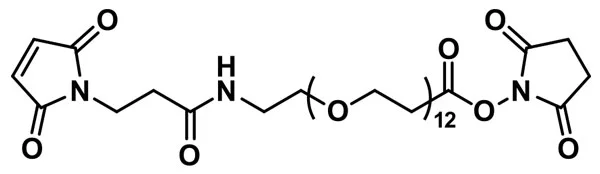
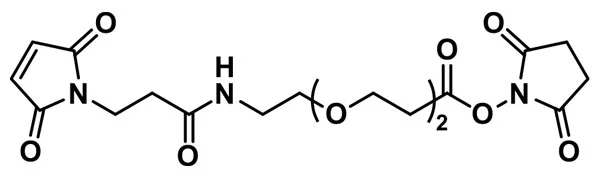
MAL-dPEG®₁₂-NHS ester
MAL-dPEG®12-NHS ester, product number QBD-10284, is a crosslinking reagent that joins a sulfhydryl to a free amine through a hydrophilic bridge. The sulfhydryl groups react with a maleimide group via a Michael addition reaction. The amines form amide bonds with the crosslinker by nucleophilic substitution of the N-hydroxysuccinimidyl (NHS) ester of a carboxylic acid group. The maleimide and NHS functional groups on the crosslinking compound sit at either end of a long, discrete-length polyethylene glycol chain (dPEG®).
Reactions that join free amines with free thiols are among the most popular, most useful crosslinking reactions in bioconjugate chemistry. These reactions require heterobifunctional reagents that bridge the two groups. Traditional crosslinkers are hydrophobic molecules. Vector Laboratories’ dPEG® crosslinking products are water-soluble, amphiphilic, single molecular weight PEG compounds with discrete chain lengths.
The conjugation of conventional hydrophobic crosslinking reagents to biomolecules almost inevitably triggers problems such as aggregation and precipitation of the conjugates. These problems do not occur with our water-soluble, non-immunogenic dPEG® crosslinkers.
Because NHS esters hydrolyze readily in water or aqueous buffer, the NHS ester end of the molecule must conjugate to a target molecule before conjugating the maleimide end of the molecule. At pH 7.0 – 7.5, NHS esters react optimally with free amines. However, NHS esters can react with free amines with pH as low as 6.5. As the pH increases, the hydrolysis rate of the NHS ester increases. Indeed, at pH 8 and 25°C, the half-life of an NHS ester in aqueous media is one hour, while at pH 8.6 and 4°C, the half-life falls to ten (10) minutes.[3] Thus, we strongly discourage storing MAL-dPEG®12-NHS ester, product number QBD-10284, in water or aqueous buffer. Instead, we recommend that customers make new solutions of the product as needed, use them immediately, and discard unused solutions after use.
The reaction of the maleimide end of MAL-dPEG®12-NHS ester with a sulfhydryl proceeds optimally at pH 6.5 – 7.5. Use the lowest reasonable pH within this range. Above pH 7.5, free amines compete with free thiols at the maleimide reaction site, which can cause confusing results. Moreover, at higher pH values, the maleimide ring may open to form unreactive maleamic acid.
The use of MAL-dPEG®12-NHS ester, product number QBD-10284, has been published in numerous scientific paper and patents.
The following list highlights some of the more important uses of this product:
- development of various imaging applications;
- development of antibody-drug conjugates and extracellular antibody-drug conjugates;
- modification of diabodies and bispecific antibody fragments;
- development of different types of assays for peptides, proteins, and oligonucleotides;
- attachment of antibodies to atomic force microscopy probes;
- creation of a scaffold for HIV inhibitors;
- development of biosensors; and
- surface coating of nanoparticles.
Also, in a radioimaging application, MAL-dPEG®12-NHS ester demonstrably improved both the biodistribution and image quality of a DOTA-conjugated diabody.
Price range: $246.00 through $711.00
| SKU | Unit Size | Price |
|---|---|---|
Select a unit size:
How do I request a quote or bulk pricing?
Documents |