dPEG®4-SATA, product number QBD-10181, is a hydrophilic, water-soluble version of the highly popular thiolation and crosslinking reagent known as SATA. Using straightforward, easy reactions, this reagent can be used to thiolate or crosslink biomolecules.
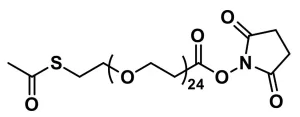
dPEG®4-SATA, also known as S-acetyl-dPEG®4-NHS ester, product number QBD-10181, is one of Quanta BioDesign’s versions of the widely popular thiolation reagent N-succinimidyl-S-acetylthioacetate (SATA). The acetyl-protected thiol is separated from the N-hydroxysuccinimidyl (NHS) ester by an amphiphilic, discrete polyethylene glycol (dPEG®) spacer. The dPEG® spacer adds water solubility to the product.
Thiolation is the process of adding a sulfhydryl group to a molecule. Bioconjugation frequently employs thiolation because the reactions to install thiol groups on molecules or to react molecules with sulfhydryl groups are simple and often chemoselective.
The widely popular SATA reagent thiolates molecules with available amines via the NHS ester. Removal of the acetyl protecting group from the sulfhydryl permits crosslinking between the SATA-modified compound and a target molecule of interest that contains a thiol-reactive group, such as maleimide. Unfortunately, SATA is hydrophobic. Consequently, before use, SATA must be dissolved in a dry, water-miscible organic solvent.
Vector Laboratories’ dPEG®4-SATA inserts a single molecular weight tetraethylene glycol (dPEG®4) spacer between the protected thiol and the NHS ester. The dPEG® spacer imparts water solubility to the molecule, allowing dPEG®4-SATA to be dissolved and reacted in water without the need for an organic solvent. Also, the dPEG®4 spacer adds hydrodynamic volume to the molecule to which it is conjugated.
Increasing the hydrodynamic volume reduces aggregation and precipitation of proteins conjugated to dPEG®4-SATA. Hydroxylamine hydrochloride (CAS number 5470-11-1) easily removes the acetyl protecting group, exposing the sulfhydryl moiety for further reaction. Any application that can be carried out with a traditional, non-PEGylated SATA reagent can be carried out with our SATA containing a dPEG® spacer.
| Unit Size | 100 mg, 1000 mg |
|---|---|
| Molecular Weight | 421.46; single compound |
| Chemical formula | C₁₇H₂₇NO₉S |
| CAS | 937025-17-7 |
| Purity | > 98% |
| Spacers | dPEG® Spacer is 16 atoms and 18.3 Å |
| Shipping | Ambient |
| Typical solubility properties (for additional information contact Customer Support) | Methylene chloride, Acetontrile, DMAC or DMSO. |
| Storage and handling | -20°C; Always let come to room temperature before opening; be careful to limit exposure to moisture and restore under an inert atmosphere; stock solutions can be prepared with dry solvent and kept for several days (freeze when not in use). dPEG® pegylation compounds are generally hygroscopic and should be treated as such. This will be less noticeable with liquids, but the solids will become tacky and difficult to manipulate, if care is not taken to minimize air exposure. |
Applicable patents and legal notices are available at legal notices.
Stay in the Loop. Join Our Online Community
Products
Ordering
About Us
Application
Resources

©Vector Laboratories, Inc. 2025 All Rights Reserved.
To provide the best experiences, we use technologies like cookies to store and/or access device information. Consenting to these technologies will allow us to process data such as browsing behavior or unique IDs on this site. Not consenting or withdrawing consent, may adversely affect certain features and functions. Privacy Statement
How do I Request a Quote?
To request a quote for products: