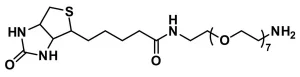
Biotin-dPEG®23-NH2 (Biotin-dPEG®23-amine), product number QBD-10786, is a carboxylate-reactive biotinylation product on a long (71 atoms), single molecular weight, discrete PEG (dPEG®) spacer. Biotin-dPEG®23-NH2 reacts with activated esters (N-hydroxysuccinimide, 2,3,5,6-tetrafluorophenyl) of carboxylic acids under slightly basic conditions. Moreover, it can be coupled directly to a carboxylic acid using 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) chemistry. Additionally, the product reacts with aldehydes or ketones to form a labile Schiff base. The Schiff base can be reduced to a secondary amine using sodium cyanoborohydride if additional conjugate stability is necessary.
Amphiphilic Biotin-dPEG®23-NH2 dissolves equally well in both aqueous media and organic solvents. Therefore, it is unnecessary to dissolve this product in an organic solvent before using it in an aqueous reaction. Furthermore, biotinylation of biomacromolecules with Biotin-dPEG®23-NH2 will not cause aggregation and precipitation. Eliminating aggregation and precipitation improves the signal-to-noise ratio of applications developed with this product. Reactions with carboxylic acids or active esters of carboxylic acids yield stable amide bonds. Aldehydes are not commonly found in biomacromolecules but can be formed by oxidizing reducing sugars in glycosylated proteins. Reactions with aldehydes create labile imines (Schiff bases) that can be reduced under mild conditions with sodium cyanoborohydride to form more stable secondary amines.
Biotin-dPEG®23-NH2 works in any application that can take advantage of the exceptionally strong biotin-avidin/streptavidin affinity. The terminal amine of QBD-10786 offers the opportunity (1) to biotinylate the carbohydrate coat of glycoproteins by oxidizing reducing sugars to aldehydes and (2) to biotinylate surfaces consisting of carboxylic acids or their active esters. Thus, this product expands the range of molecules and nanoparticles in which dPEG®-biotin can be employed.
| Unit Size | 100mg, 1000mg |
|---|---|
| Molecular Weight | 1299.60; single compound |
| Chemical formula | C₅₈H₁₁₄N₄O₂₅S |
| CAS | 604786-74-5 |
| Purity | > 95% |
| Spacers | dPEG® Spacer is 71 atoms and 87.0 Å |
| Shipping | Ambient |
| Typical solubility properties (for additional information contact Customer Support) | Methylene chloride, DMAC, DMSO or water. |
| Storage and handling | -20°C; Always let come to room temperature before opening; be careful to limit exposure to moisture and restore under an inert atmosphere; stock solutions can be prepared with dry solvent and kept for several days (freeze when not in use). dPEG® pegylation compounds are generally hygroscopic and should be treated as such. This will be less noticeable with liquids, but the solids will become tacky and difficult to manipulate, if care is not taken to minimize air exposure. |
Greg T. Hermanson, Bioconjugate Techniques, 2nd Edition, Elsevier Inc., Burlington, MA 01803, April, 2008 (ISBN-13: 978-0-12-370501-3; ISBN-10: 0-12-370501-0); See pp. 529-530 for a general discussion in Greg’s book and on biotinylation as well, as well as pp. 737-738 for a protocol for a compound with just two oxygen. However, other than enhanced solubility and binding distances, the protocol is adaptable to our PN 10193.
Greg T. Hermanson, Bioconjugate Techniques, 3rd Edition, Elsevier, Waltham, MA 02451, 2013, ISBN 978-0-12-382239-0; See Chapter 18, Discrete PEG Reagents, pp. 787-821, for a full overview of the dPEG® products.
Blood Plasma-Derived Anti-Glycan Antibodies to Sialylated and Sulfated Glycans Identify Ovarian Cancer Patients. Tatiana Pochechueva, Alexander Chinarev, Andreas Schoetzau, Andre Fedier, Nicolai V. Bovin, Neville F. Hacker, Francis Jacob, Viola Heinzelmann-Schwarz. Plos One. 2016, 11 (10) e0164230. October 20, 2016. DOI: 10.1371/journal.pone.0164230.
Poly(catecholamine) coating and biofunctionalization of CsPbBr3 microcrystals. Sangyeon Cho and Seok Hyun Yun. ChemRxiv. 2021. License: CC BY-NC-ND 4.0. DOI: 10.26434/chemrxiv.14049710.v2
Applicable patents and legal notices are available at legal notices.
Stay in the Loop. Join Our Online Community
Products
Ordering
About Us
Application
Resources

©Vector Laboratories, Inc. 2025 All Rights Reserved.
To provide the best experiences, we use technologies like cookies to store and/or access device information. Consenting to these technologies will allow us to process data such as browsing behavior or unique IDs on this site. Not consenting or withdrawing consent, may adversely affect certain features and functions. Privacy Statement
How do I Request a Quote?
To request a quote for products: