
Vector Laboratories is closed for the President’s Day on Monday, February 19th. We will be back in the office on Tuesday, February 20th.
We will respond to emails upon our return. Have a wonderful day.
Menu
Vector Laboratories is closed for the President’s Day on Monday, February 19th. We will be back in the office on Tuesday, February 20th.
We will respond to emails upon our return. Have a wonderful day.

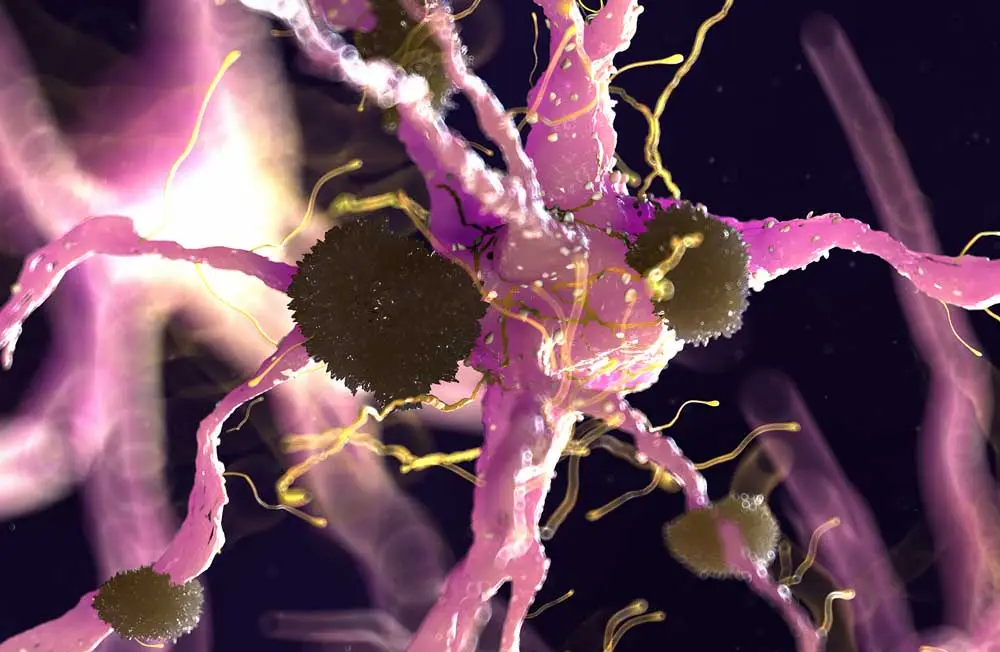
Since it was first described in 1906 by Alois Alzheimer, physicians and scientists have been searching for clues to the debilitating neurological condition Alzheimer’s disease (AD). Today, AD is the most common cause of dementia among older adults and has no known cure. Hallmarks of AD include β-amyloid plaques and neurofibrillary tau tangles that are both surrounded by activated microglia. This activation can be stimulated by various stimuli, including β-amyloid load, during neuroinflammation. To diagnose and treat the disease at its earliest stages, researchers are searching for additional biomarkers that can help guide our understanding of the disease.
Microglial activation, which is mediated by an overexpression of the soluble fragment of triggering receptor expressed on myeloid cells 2 (sTREM2), is involved in the development of AD.
TREM2, the transmembrane receptor on microglia, plays a role in phagocytosis and neuroinflammation. It is associated with total and phosphorylated tau levels (T- and P-tau), including Thr181 and Ser 199, which are implicated in tau pathology. Tau are tubulin-associated proteins that form aberrant aggregates during hyperphosphorylation [1]. Partial or normal TREM2 levels contribute to tau-mediated brain injury in a complex role during early and late-onset AD. And while dysfunctionally low levels of TREM2 may cause tau hyperphosphorylation in early stages of the disease, TREM2 loss-of-function could be beneficial in advanced stages [2]. Many studies suggest that sTREM2 is a neuroinflammatory marker of the early stages of AD, with elevated sTREM2 levels in mild cognitive impairment (MCI) being neuroprotective against inflammation in AD [3].
Since cerebrospinal fluid (CSF) sTREM2 is elevated in AD, researchers from Seongnam Center of Senior Health, Republic of Korea, focused on its relationship with other AD biomarkers, correlating plasma sTREM2 with CSF sTREM2 in groups with or without AD [4]. Their specific aim was finding a relationship between multiple biomarkers and elucidating the pathophysiology of this debilitating disease.
Biomarker analysis of CSF sTREM2 positively correlated with CSF T-tau and P-tau in the amyloid-positive group. CSF sTREM2 was also strongly correlated with neurofilament light chain, which is significant as elevated neurofilament light chain may result in axonal damage from neuroinflammation, especially in multiple sclerosis. High sTREM2 levels associated with lower levels of P-tau in the early-onset amyloid-positive group. Together, these results demonstrate that sTREM2 can be a potential biomarker of AD.
CSF sTREM2 levels had an inverse correlation with plasma sTREM2, which is discrepant in the literature. Some papers reported a significant association between peripheral sTREM2 and CSF sTREM2 in AD patients, but other studies reported no difference in plasma sTREM2 between AD and healthy groups. Increased levels of plasma sTRME2 in the elderly is consistent with its potential role as microglial marker. Initially, decreased levels of CSF P-tau and T-tau in the elderly was contradictory when CSF sTREM2 and tau levels were positively correlated. Further analysis found that earlier onset (< 65 years) AD patients were overrepresented in the study, producing an incorrect result. Amyloid-positive groups had slightly lower but statistically insignificant CSF Aβ42 (amyloid-β) levels, but other studies found that CSF P-tau 181 has been validated with T-tau and Aβ42 as AD biomarkers [5].
Researchers detected sTREM2 in CSF and plasma using solid phase Proximity Ligation Assay (spPLA), which improves the limit of detection with surface-bound capture antibody. Streptavidin-oligonucleotide conjugates were formed using the Protein-Oligo Conjugation Kit featuring SoluLINK® bioconjugation technology. With an immobilized capture antibody, proximity probes were oligonucleotides synthesized with streptavidin at the 5’ or 3’-end, coupled with biotinylated antibodies. These proximity probes then detected the captured target sTREM2 in blood plasma and CSF. Magnetic beads were incubated with biotinylated anti-sTREM2 polyclonal antibody to immobilize sTREM2 antibodies to beads. Recombinant TREM2 protein was serially spiked in PLA buffer as a standard and mixed with antibody-conjugated magnetic beads. PLA probes were formed by incubating streptavidin-oligonucleotide conjugates (SLC1 and SLC2) with biotinylated anti-sTREM2. Finally, the magnetic beads were mixed with PLA probe mix. Real-time PCR was performed for quantification of the ligated oligonucleotide sequences, which were joined together by a splint primer.
ELISA was used to measure plasma and CSF biomarker levels. A human p-tau S199 detection antibody was applied to wells with HRP anti-rabbit IgG secondary antibody. Then, TMB substrate was added, the reaction was halted with stop solution, and optical density was read at 450 nm. To measure total-tau in CSF, biotinylated tau antibodies were detected with streptavidin HRP and TMB substrate.
Telomere length was assayed as a biomarker to correlate with P-tau and NfL in linear regression analysis. DNA extracted from whole blood was analyzed by southern blot, and DNA fragments were hybridized to digoxigenin (DIG)-labeled probes specific for telomeric repeats. The nylon membrane was then incubated with alkaline phosphatase — conjugated anti-DIG antibody. Unconjugated anti-DIG can be labeled with alkaline phosphatase using a Protein-Protein Conjugation Kit which also features SoluLINK bioconjugation technology.
sTREM2 is a potential biomarker of microglial activation and subsequent neuroinflammation in AD. A change in microglial activation and clustering around β-amyloid plaques, which is mediated by sTREM2, is implicated in AD pathophysiology. Additionally, analysis of CSF and plasma in individuals with and without AD suggests that elevated levels of sTREM2 may lead to tau-induced neurodegeneration in AD.
Several limitations should be noted, however, including a lack of statistical power from a small sample size (n = 104) across amyloid status groups, though this may be mediated in the future by a large-scale study. Researchers did not follow up on biomarkers to observe long-term changes in sTREM2 levels during the progression of AD. And, the choice of P-tau S199 for tau hyperphosphorylation status could be replaced by more commonly characterized P-tau181 or P-tau217 markers. A longitudinal study using these markers may validate current data on sTREM2’s role in AD and clarify conflicting findings from previous investigations.
References




Stay in the Loop. Join Our Online Community
Together we breakthroughTM

©Vector Laboratories, Inc. 2024 All Rights Reserved.