
Vector Laboratories is closed for the President’s Day on Monday, February 19th. We will be back in the office on Tuesday, February 20th.
We will respond to emails upon our return. Have a wonderful day.
Menu
Vector Laboratories is closed for the President’s Day on Monday, February 19th. We will be back in the office on Tuesday, February 20th.
We will respond to emails upon our return. Have a wonderful day.
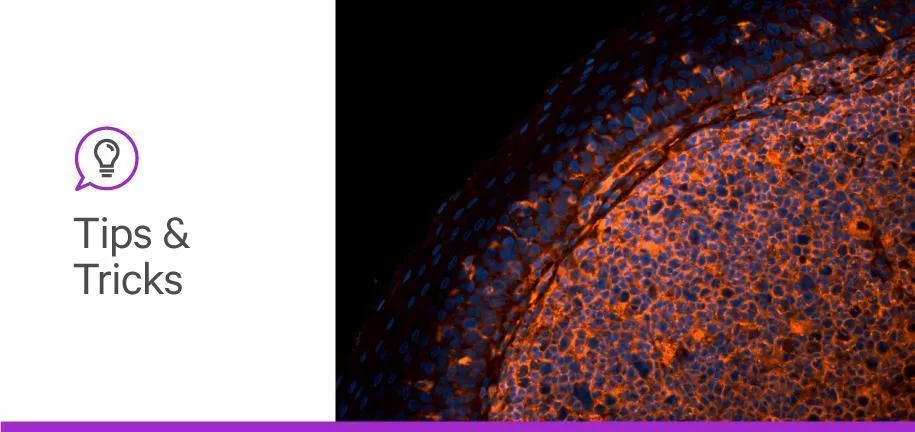
Understanding the role of glycans in cellular processes and pathology is necessary to unlock scientific breakthroughs. The glycan profiling of a cell population can be quite informative in both healthy and diseased states. Not only can we learn about the roles of glycans in essential bodily functions, but we can also deduce how alterations in glycan profiles modulate disease phenotypes. These findings, when combined, can significantly contribute to drug discovery.
For example, tumor cell surface glycome could inform scientists on how to design targeted therapeutics with higher affinity to tumor cells. The only setback is the lack of knowledge about the involvement of glycans in health and diseases. The gap can only be closed by investigating patient samples to reveal the amount, dynamics, and location of glycan structures. This can be achieved by glycan-binding proteins, also called affinity reagents: glycan-binding antibodies and lectins. The former is generated by the body or the cell population as an immune response to a foreign glycan, while the latter directly binds specific glycans at its recognition domain.
By quantifying and visualizing antibody or lectin binding, researchers can gain valuable insight into the glycan profile of the sample. As mentioned in our previous article on lectins, the vast diversity and unique physicochemical properties of lectins make them ideal candidates for glycan detection.
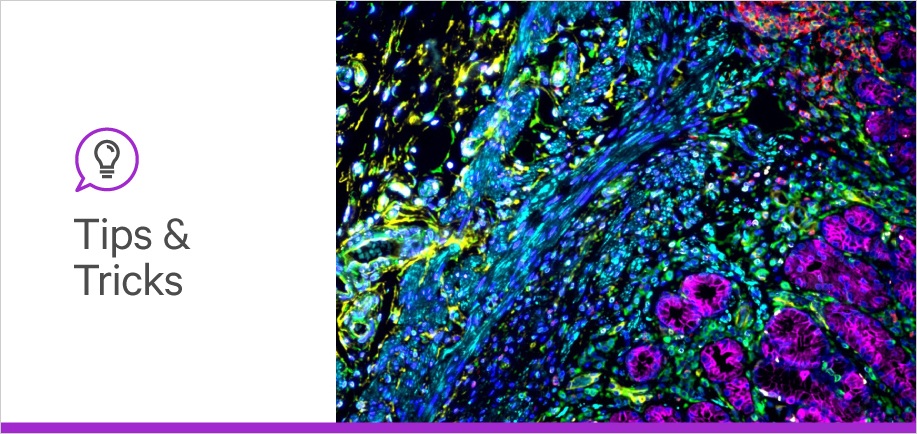
Although the mechanism of binding slightly differs between glycan-binding proteins, their employment of glycan-detection assays, including immunohistochemistry (IHC) and immunofluorescence (IF), is quite similar. These two methods are necessary for visualizing and quantifying glycan structures in the sample.
This article summarizes the considerations for using lectins in glycobiology assay workflows. Although many steps are identical to those in the antibody-integrated workflows, there are subtle but crucial differences.
Preserving the tissue is the first step to a successful IHC/IF design. Regardless of what affinity reagent you use, tissue preparation can be achieved by either freezing or paraffin embedding.
Paraffin embedding involves dehydration (fixation) of the tissue in alcohol before embedding in paraffin, which hardens overnight and allows the tissue to be stored at room temperature for much longer. On the other hand, freezing does not involve the initial fixation, and the tissue is instead immersed in liquid nitrogen or dry ice. This method is often preferred for retaining enzymatic activity.
Tissues often contain endogenous peroxidase, pseudoperoxidase, and alkaline phosphatase (AP). This predisposes them to background staining when peroxidase and AP detection systems are used. Quenching can mitigate the risks by inactivating these enzymes with a dual enzyme-blocking reagent, such as BLOXALL® Endogenous Blocking Solution.
Blocking is where antibody- and lectin-integrated workflows start to differ.
In antibody-based workflows, biotin labeling is used to amplify the ability of antibodies to detect antigens. Due to its high affinity for biotin, a glycoprotein called avidin is commonly used to leverage avidin-biotin binding to amplify staining intensity in IHC. However, tissues may contain endogenous biotin, which leads to nonspecific background staining. This can be prevented through pre-treatment with avidin/biotin-blocking reagents.
Although avidin has advantages such as high solubility and low production costs, its use in lectin-based workflows is not recommended. Because avidin is a glycoprotein itself, it could form nonspecific interactions with lectins and alter the final staining. Streptavidin is an alternative that could be used for blocking biotin. Due to its lack of carbohydrate side chains, nonspecific binding is significantly reduced.
Another source of nonspecific staining comes from the interactions between the primary antibody and Fc receptors. In antibody-based IHC workflows, one can use normal sera, a protein block obtained from healthy animal milk, preferably the species used to raise the secondary antibodies. By nonspecifically binding Fc receptors in the sample, normal sera prevents the primary antibodies from binding.
However, normal sera milk is not suitable for lectin workflows for the same reason avidin is avoided. Not only normal sera milk but also all other milk proteins are highly glycosylated; therefore, they cause undesired lectin binding.
Bovine Serum Albumin can be substituted as the blocking reagent in lectin-based workflows. Another alternative is the Carbo-free blocking solution. It is free of glycoproteins, which mitigates the risk of background staining and subsequent false positives.
A primary antibody directly binds the target protein at its moiety. For lectin workflows, a glycan-specific lectin can be used instead of the primary antibody. Regardless of the choice of the primary binding reagent, you need to consider the constituents of your tissue/sample, as well as the tissue preparation and blocking methods to ensure the optimum level of specific binding.
Secondary reagents are used to amplify the signal intensity and confer additional properties to the primary antibody, such as immunolabeling.
If the primary reagent is an antibody, anti-antibodies such as anti-human immunoglobulin antibodies must be used. These antibodies can be found in unconjugated or conjugated formats. Unconjugated secondary antibodies are ideal for custom conjugation, while commercially available conjugated antibodies enable amplified signaling through fluorophores or biotin labeling.
When primary lectins are used instead of antibodies, anti-lectin becomes the ideal secondary reagent. If the lectin is biotinylated, streptavidin must be used as the anti-lectin. If the primary lectin itself has been tagged, anti-lectin antibodies, such as anti-fluorescein, anti-DNP, anti-DIG, and anti-biotin, can be used.
Tertiary reagents are useful for signal amplification in low-abundance molecules of interest.
In IHC workflows, tertiary reagents and chromogens are used to bind the secondary antibody to form a chromogenic product. Tertiary reagents often comprise enzymes, such as horseradish peroxidase (HRP) and alkaline phosphatase (AP), which interact with the chromogens/substance, which leads to enzyme-specific color development. This interaction is often used to produce crisp staining that reveals extracellular location and relative expression levels of molecules of interest.
IF workflows involve amplified fluorescent systems, where an amplifier antibody (e.g., Anti-Mouse IgG or Anti-Rabbit IgG antibody produced in goats) binds the secondary reagent. Then, a dye-conjugated Anti-Goat IgG antibody is attached to the amplifier antibody to yield bright fluorescence staining.
Integrating lectins into IHC/IF workflows is not as difficult as it may sound. In fact, many steps in the beginning and the end are nearly the same. The only critical considerations for lectin usage are the choice of blocking reagents and secondary detection reagents, which would amplify your signal and eliminate background staining.
If you are unsure whether to choose antibodies or lectins for glycan detection, check out our article highlighting the key advantages of lectins.
Other resources include the following articles and databases. UniProt is a reliable protein database to check if a protein to be integrated into the workflow is glycosylated. An optimized protocol for combined fluorescent lectin/immunohistochemistry to characterize tissue-specific glycan distribution in human or rodent tissues summarizes the rationale and considerations behind lectin-based IF/IHC methods. PubMed houses many more articles demonstrating lectin use in specific applications.
For those wanting to expand their knowledge of lectins, Vector Laboratories offers The Lectins Application and Resource Guide, packed with valuable information from lectin applications—including the Coronavirus research—to sugar specificity.




Stay in the Loop. Join Our Online Community
Together we breakthroughTM

©Vector Laboratories, Inc. 2024 All Rights Reserved.