
Vector Laboratories is closed for the President’s Day on Monday, February 19th. We will be back in the office on Tuesday, February 20th.
We will respond to emails upon our return. Have a wonderful day.
Menu
Vector Laboratories is closed for the President’s Day on Monday, February 19th. We will be back in the office on Tuesday, February 20th.
We will respond to emails upon our return. Have a wonderful day.


As a child, Andrea Jeffers Wellington was fascinated with all the life forms in the ocean and secretly wished that all the water would disappear so that she could walk on the sea floor and observe all the cool creatures. She nurtured hamsters and snakes as pets along the way and found ways to learn more about them and their biology as she matured. Like many in the sciences, she loved science lab in high school and chose collegiate classes, like animal physiology, that ultimately served her well across her thirty-year science career. Today she works in a research lab at the University of Arizona that studies diabetic retinopathy, hopeful that this work will ultimately lead to solutions that will help prevent sight-threatening damage.
“I started my scientific career using molecular biology to study cytoskeletal proteins and proteins that bind to them. Then I moved to a neuroscience lab where I used molecular biology and biochemistry to study a gene involved in mitochondrial transport…I moved to my current lab where I continue doing confocal microscopy and image analysis in the study of diabetic retinopathy in mice.”
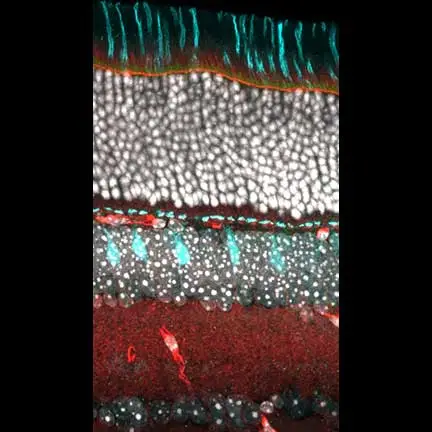
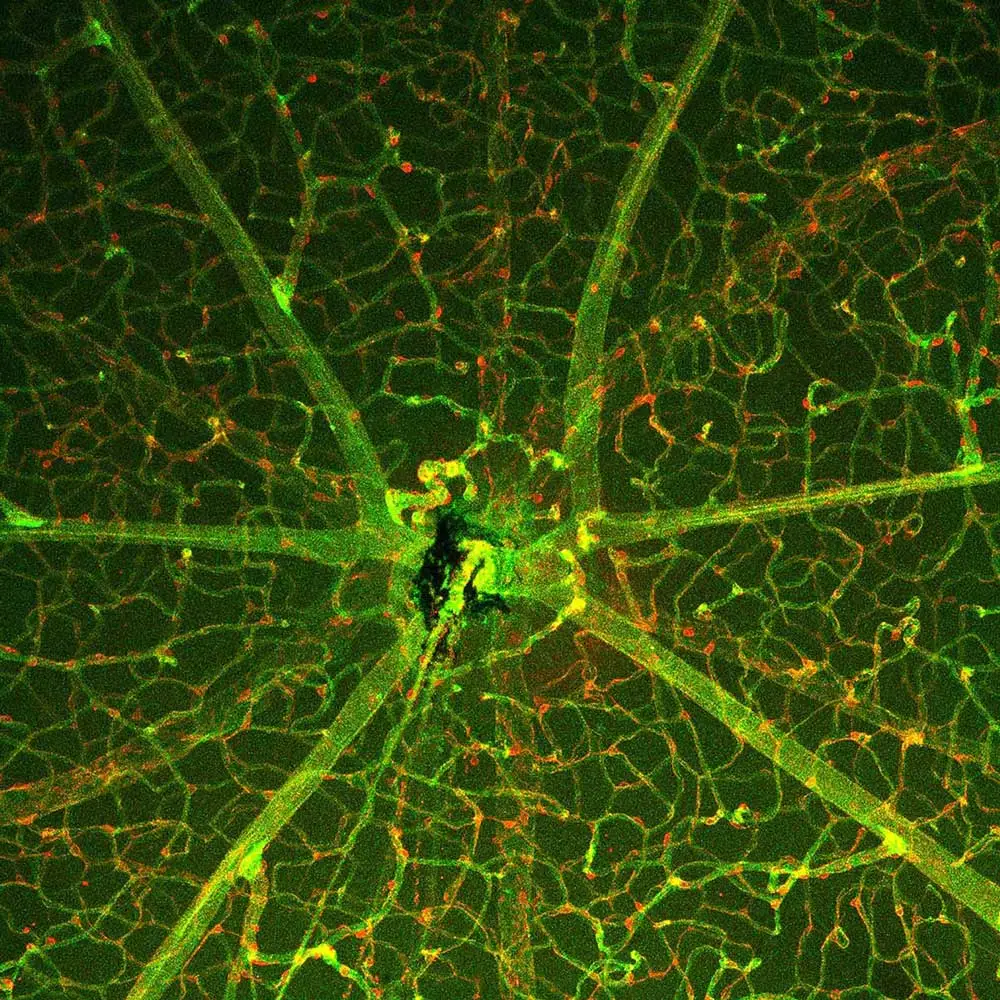
Above Right Image: Slice from mouse retina tissue showing cone bipolar cells genetically labeled in cyan, cones and their processes also labeled in cyan (stained with peanut agglutinin-fluorescein), actin labeled in red, and cell nuclei labeled in white.
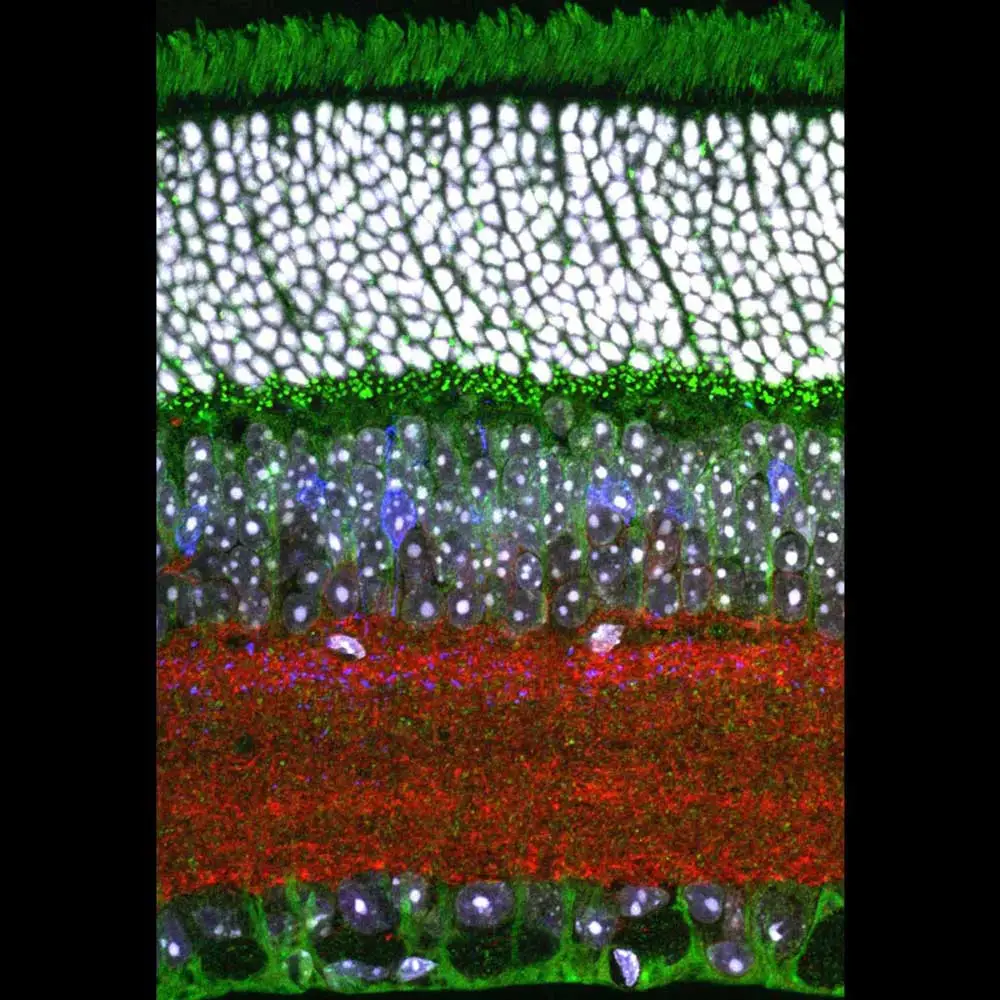
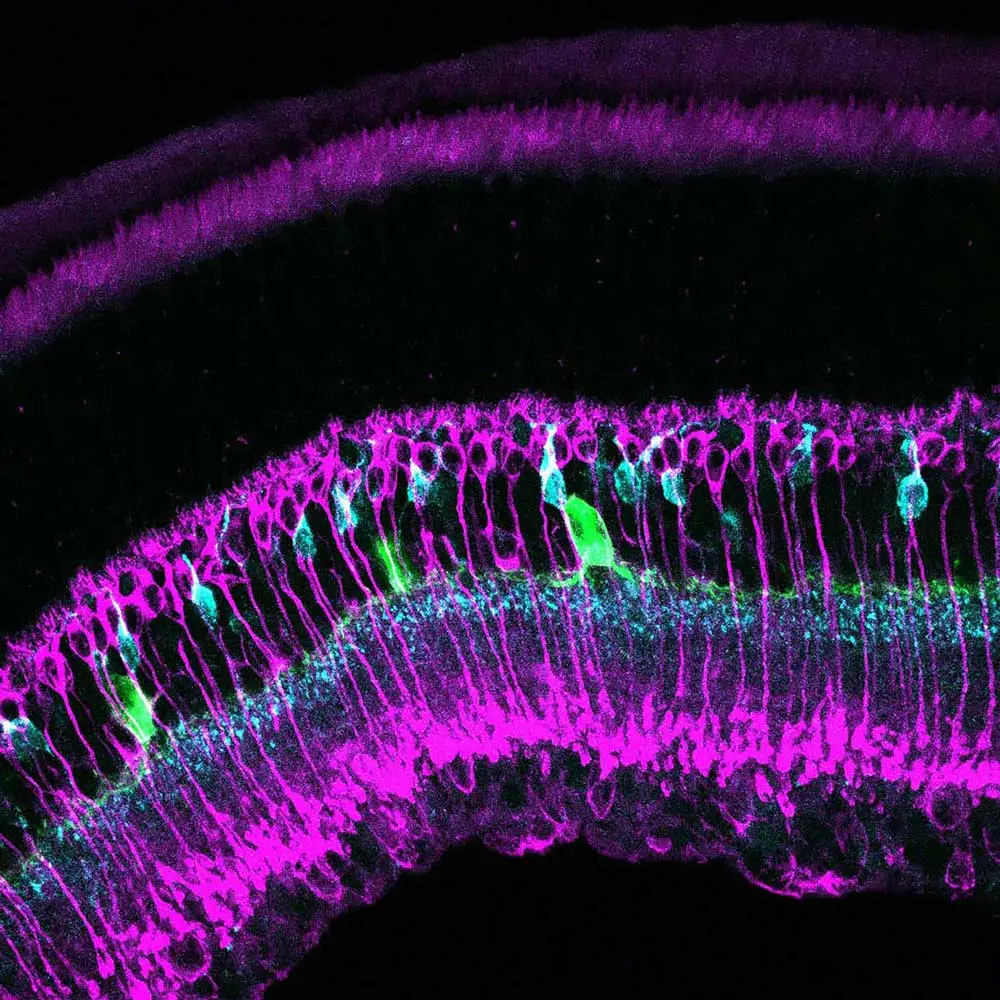
Andrea describes the way that she came into her current area of expertise as a fortuitous sequence of events that provided opportunities to learn and apply new techniques. With microscopy, she found that being able to watch something that no one had ever observed before—like mitochondria moving along neurons in mutant fruit flies that she created—was very intriguing. The lab she works in now uses immunostaining and analysis, which can be tedious at times, to help quantify retinal differences between the diabetic and control mice. What makes up for the tedium, in addition to the potential to help diabetic patients retain their sight, is that the images created during this process can be quite beautiful. So much so that she’s turned a number of these images into artwork and entered the images into photo contests, yearly biological art shows organized by the Neuroscience Club at her university, and shared them with her lab mates as prints on T-shirts, mouse pads, and other functional objects (including facemasks).
“COVID-19 has affected my research in that I’m now doing all my image analysis from home. Our lab members stagger our schedules so that only one of us is in the lab at a time. I go into the lab only as needed to inject mice and do their weekly urine glucose tests.”
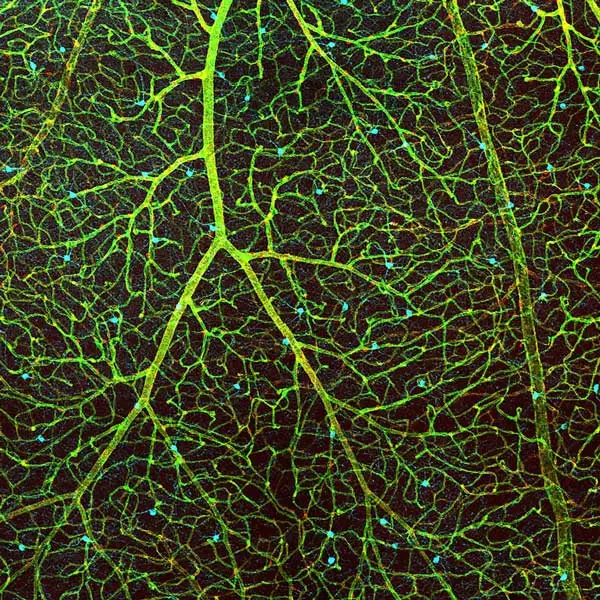
One of disappointments resulting from the coronavirus situation is that for the first time in four years, she could not mentor a student in the Keep Engaging Youth in Science (KEYS) program. This is a free research internship program for motivated high school students that offers them credit and valuable experience. When she can provide mentoring, her best advice for students doing immunostaining is to validate new antibodies in any model before using them in your experiment, understand that the antibody used in someone else’s publication might not look the same in the tissue or organism of interest to you, and be sure to use a negative control (knockout) to determine the specificity of the antibody you choose.
“I do find science progress to sometimes be inherently slow. When I leave the lab, I choose activities that have a much different pace, like motorcycling, or where I can achieve goals quicker, like weightlifting. For example, in the 2020 US Powerlifting Association competition held in August, I set a world record in the bench press, set personal records in the squat and deadlift events, and earned 4 gold medals. It was fun.”
When moving from one research area to another over the years, Andrea initially found challenges in moving from using invertebrate models like worms and fruit flies to a vertebrate model using mice. Part of that learning curve involved learning how to master mouse handling skills, but also how to manage a paradigm shift, as the mindset in making transgenic worms or flies, which can be done quickly and inexpensively, is different than the one needed to generate transgenic mice, complicated by the specific protocols that must be followed and ending up being significantly more expensive and time-consuming. Having made these kinds of transitions more than once along the way, Andrea considers herself to be really lucky, because she enjoys doing the hands-on work in the lab and is enthusiastic to see what path science will take her.




Stay in the Loop. Join Our Online Community
Together we breakthroughTM

©Vector Laboratories, Inc. 2024 All Rights Reserved.