An Introduction to the Universe of Glycans

From unicellular organisms to mammals, all living things owe their functionality and integrity to 4 building blocks: nucleic acids (DNA and RNA), proteins, lipids, and polysaccharides, also known as glycans. Among these macromolecules, glycans are perhaps the most mysterious due to their dazzling structural diversity and various functions. In fact, the more we learn about glycans, the more we realize that we are still scratching the surface of glycobiology. To add to that, it is impossible to think of glycans in isolation. They are composed of glycosidically linked monosaccharides conjugated to other macromolecules. That’s why understanding the fundamentals of glycans can pave the way for elucidating their roles in essential biological processes.
Where Does Glycan Structural Diversity Come From?
To understand the factors behind glycan structural diversity, we need to go back to their building blocks: monosaccharides. More specifically, the answer lies in how 2 monosaccharides join together by a glycosidic bond. Unlike amino acids and nucleotides, with a more standardized coupling to form proteins and nucleic acids, 2 monosaccharides can bind at several locations, resulting in the possibility of several isomeric structures. To add to that, 1 monosaccharide can form glycosidic linkages at multiple locations, giving rise to a structural phenomenon not encountered in other macromolecules: branching. As the size of a polysaccharide grows, the number of possible structures increases exponentially.
Structural Classification of Glycans
Glycans are conjugated with proteins and lipids to confer specific structures and functions, such as protein folding, recognition, and cell migration. Despite the staggering diversity of glycans, a high-level classification is possible depending on the conjugation type and the binding site.
Glycoproteins
The covalent bonding of a glycan to a protein, glycosylation, is one of the most common posttranslational modifications in eukaryotic proteins. Unlike the diverse nature of glycosidic linkages between monosaccharides, glycosylation is more constrained since glycans can bind peptides only at the binding sites composed of specific amino acid sequences. These glycans can be classified according to the atom on the amino acid residue side chain at the binding site.
N-Linked glycans
N-linked glycans are attached to the nitrogen of the asparagine side chain, where Asn is part of either Asn-X-Ser or Asn-X-Thr, provided that X is any residue but proline.
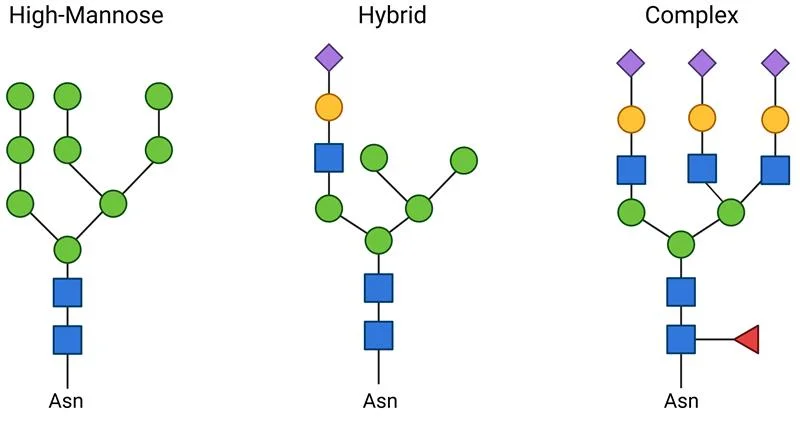
The precursor structure of an N-glycan can contain 1 of the following monosaccharides: N-acetylgalactosamine, galactose, neuraminic acid, N-acetylglucosamine, fucose, and mannose. That said, the precursor core structure of an N-glycan is conserved. The precursor oligosaccharide is formed in the ER, starting with 2 N-acetylglucosamine (GlcNAc) residues, followed by the subsequent addition of 9 mannose and 3 glucose residues. This core glycan structure is transferred to the luminal side of the ER, where it forms a bond with the polypeptide and has its glucose residues removed.
The diversification of N-glycans occurs in cis-Golgi with the help of glycosyltransferases and glycosidases that can add and remove sugar residues, respectively. This gives rise to 3 main N-glycan types:
- High-mannose glycans have 2 N-acetylglucosamines linked to branches consisting of several mannose residues.
- Complex N-glycans have branches that contain other types of monosaccharides besides mannose.
- Hybrid N-glycans have both high-mannose and complex branches.
O-Linked glycans
O-linked glycans are oligosaccharides attached to the oxygen atom of the side chains of Serine or Threonine residues. We can further classify O-glycans according to the sugar residue attached to the peptide.
In many O-glycosylated proteins, mainly membrane glycoproteins, the first sugar to attach is N–acetyl-galactosamine (GalNAc). Although there is no specific residue sequence preference for glycosylation, as there are several GalNAc transferases with different site preferences, O-glycosylation sites are often close to a proline residue.
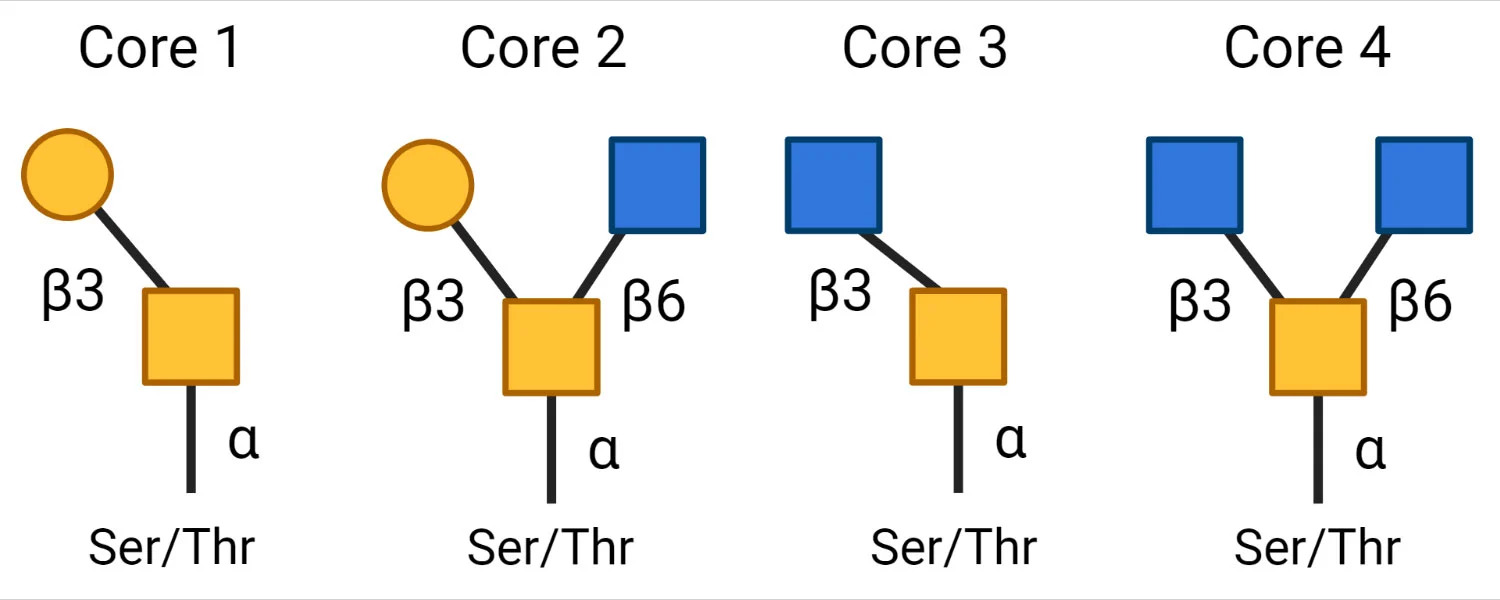
Subsequently, different core structures are possible depending on the next monosaccharide. The 4 most common core structures are as follows:
- Core 1: Addition of galactose
- Core 2: Addition of GlcNAc to the GalNAc of the Core 1 structure
- Core 3: GlcNAc addition to the first GalNAc (before Core 1 formation)
- Core 4: A second GlcNAc is added to Core 3.

Sugar residues, such as galactose, GlcNAc, sialic acid, and fucose, can be added to the core structures by glycosyltransferases.
Other O-glycans include O-GlcNAc, O-Mannose, O-galactose, and O-glucose, although they are less common than O-GalNAc. In particular, O-GlcNAc is found on cytoplasmic and nuclear proteins in the cell. The O-glycosylation of intracellular proteins is less diverse than that of extracellular glycoproteins. One theory is that extracellular glycosylation evolved to be more diversified to facilitate the binding of various pathogens and glycan-binding proteins.
Glycosaminoglycans (GAGs)
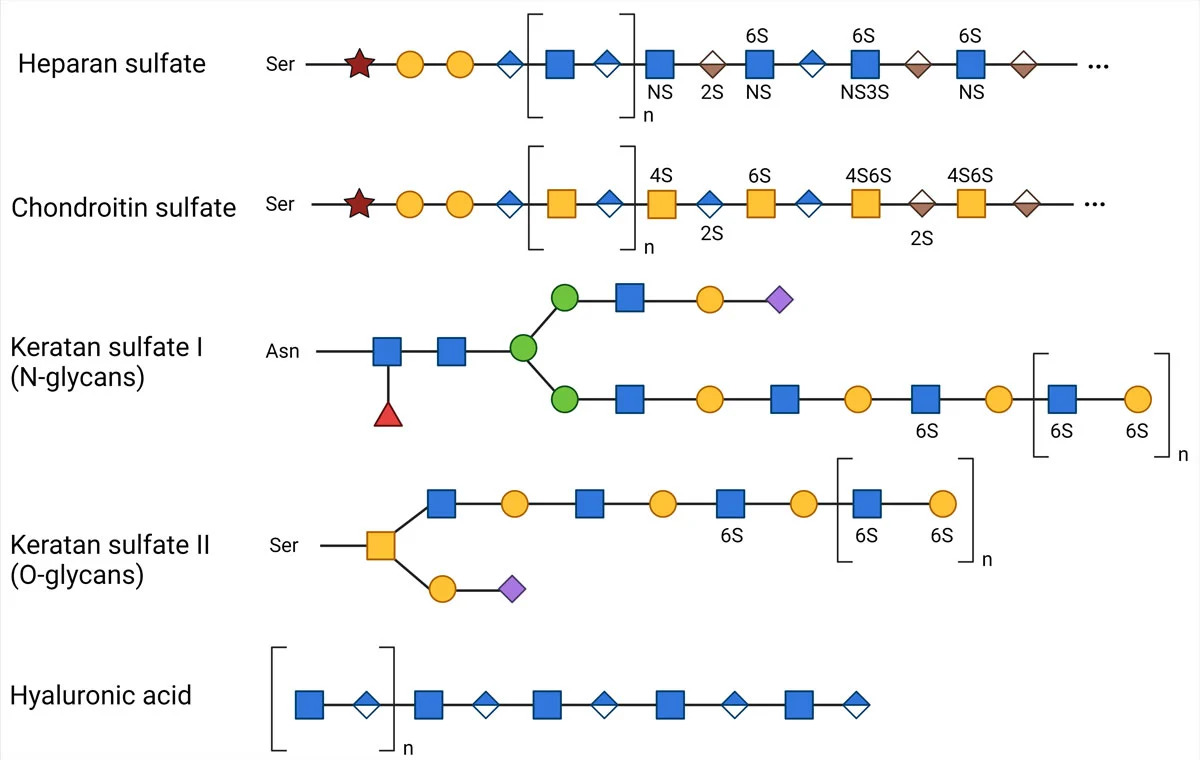
Glycosaminoglycans are long and linear polysaccharides with a repeating disaccharide unit, where a galactose residue or a uronic sugar (containing carbonyl and carboxylic acid groups, e.g., glucuronic acid or iduronic acid) is attached to an amino sugar (where 1 hydroxyl group is replaced with amine -NH2, e.g., GlcNAc or GalNAc).  There are 4 classes based on their repeating core units, with each type involving a different synthesis mechanism.
- Heparin: GlcNAc + hexuronic acid
- Chondroitin Sulfate: GalNAc + hexuronic acid
- Keratan sulfate: Galactose + GlcNAc
- Hyaluronic Acid: Glucuronic acid + GlcNAc

The synthesis of GAGs starts in the cytoplasm, where 5 uridine diphosphate (UDP) derived active sugars are formed. With the exception of Hyaluronic Acid, all GAG precursors undergo sulfation in their functional groups, which takes place in the Golgi apparatus. In contrast, the precursor sugars of Hyaluronic Acid are transferred directly to the cell membrane.
Glycolipids
Besides proteins, glycans can also be covalently attached to lipids to form glycolipids crucial to cell membrane integrity, cell-cell interactions, and immune response. Glycolipids are classified according to their hydrocarbon backbone: glycerolipids with glycerol and sphingosine with sphingolipids.
Biological Roles of glycans
It is nearly impossible to attribute a specific function to a glycan or glycan subclass. The same glycan structure can serve a range of tasks across different organisms as well as at different locations of the same organism. That’s why we present a broadened and simplified classification of glycan function.
Structural and modulatory functions
Glycans are ubiquitous in cellular compartments, membranes, and extracellular matrix, where they alter the structure and function of the proteins and lipids that they are attached to. Therefore, glycans play a critical part in shaping primary macromolecule structures, contributing to the integrity of cells and cellular components. Glycosylated macromolecules are paramount to a long list of structural and modulatory functions, from protection and transport to site-specific functions, such as lubrication of the joints to prevent friction (1).
Perhaps the most well-known example is the mucin peptide, which forms protective layers on the epithelial cell surfaces to protect tissues against pathogens and free radicals (2). In fact, aberrations in cell surface glycosylation are often associated with inflammatory diseases and cancer. Another example of a protective function is that provided by cellulose (3) and chitin (4), glycan polymers made of glucose and GlcNAc, respectively, which are indispensable components of cell walls that provide strength and stability for plants and fungi.
Glycan-induced structural modifications can also alter the physical properties of macromolecules to confer advantages. Blood plasma is the first example to come to mind since it contains a high concentration of glycoproteins, where hydrophilic glycans augment protein solubility (5). In addition, glycosylation alters the charge density of proteins, thereby preventing their degradation by proteases (6).
Extrinsic functions
Many organisms tend to form different forms of symbiotic relationships with one another, and it has become evident that cell surface glycans are one of the main control points of these interactions.
Terminal glycan residues are particularly critical to pathogen-host interactions. For instance, parasitic bacteria, including E. coli (7), and Helicobacter (8), adhere to the host cells by recognizing cell surface sialoglycans. Another example is malaria, where P. falciparum invades red blood cells by binding specific sialic acid residues on the surface (9).
Extrinsic recognition also applies to viral infections, including the most recent pandemic caused by COVID-19. A growing body of evidence suggests that N– and O-glycosylation of the spike protein strengthens its binding affinity to the ACE2 receptor while helping it evade the host immune system (10).
Fortunately, extrinsic recognition is bidirectional, meaning that host immune systems combat pathogens through glycan recognition. More specifically, glycan-binding proteins (which we will cover in the next blog post in more detail) can recognize the surface glycans of pathogens to entrap and destroy them.
Intrinsic functions
Cell-cell communication and cellular trafficking are greatly influenced by glycan recognition. Cellular uptake mechanisms, such as endocytosis and phagocytosis, are often mediated by the cell surface receptors that recognize macrophage terminal glycans. This allows cells to pull in molecules from outside the cell, transport material across organelles, and discard unwanted material.
Essential processes in organelles are also mediated by glycan receptors. For example, receptors in the ER carry out specific folding patterns for the incoming proteins based on their glycan epitopes. By the same logic, incorrectly folded proteins can be detected and discarded upon recognizing aberrant glycan structures.
Cell-cell interactions also rely on glycan recognition by surface proteins and are crucial for the survival of individual organisms and the harmony of entire ecosystems. Well-characterized examples include glycosylated fibrinogen-induced blood clotting (11) and egg-sperm interactions (12). On a larger scale, glycan-mediated intercellular signaling between rhizobacteria is largely responsible for establishing their symbiotic relationship with plants for nitrogen fixation, which has a large impact on the food chain (13).
Molecular mimicry of host glycans by pathogens
Pathogens that often evade innate immune reactions decorate their cell surfaces with glycan structures found on host cell surfaces (14). This means that pathogens can camouflage their antigenic epitopes behind these glycans. This can be achieved through various mechanisms, such as the appropriation of host glycans or convergent evolution of the pathogen, enabling the mimicry of the biosynthetic pathways of the target host.
The study of glycans can provide us valuable insights about their roles in our biological systems. While there is an overwhelming diversity in glycan structure and function, we believe that this article recaps the fundamentals of structural diversity and the functional significance of glycans.
That said, there are several resources for you to expand your knowledge of glycan biochemistry. In the article “Sweet systems: technologies for glycomic analysis and their integration into systems biology”, Chen, et al. explains glycan function with examples from the 4 classes of functions. The article also discusses the current setbacks and questions in glycobiology. For more specific carbohydrate related data, you can check out GlyGen: Computational and Informatics Resources for Glycoscience. Last but not least, our SpeakEasy Science Blog and Glycobiology eBook serve as a scientific hub where you can find practical information about glycan detection and quantification as well as reviews of peer-reviewed articles exploring the roles of glycans in cancer.
References
- Svala E., et al. 2015. Characterisation of Lubricin in Synovial Fluid from Horses with Osteoarthritis. Equine Veterinary Journal.
- Hanisch FG. 2001. O-Glycosylation of the Mucin Type. Biological Chemistry.
- McFarlane HE, et al. 2014. The Cell Biology of Cellulose Synthesis. Annual Review of Plant Biology.
- Koch BEV, et al. 2015. Keeping Track of the Growing Number of Biological Functions of Chitin and Its Interaction Partners in Biomedical Research. Glycobiology.
- Hoffmann M, et al. 2016. Site-Specific O-Glycosylation Analysis of Human Blood Plasma Proteins. Molecular & Cellular Proteomics.
- Loomes KM., et al. 1999. Functional Protective Role for Mucin Glycosylated Repetitive Domains. European Journal of Biochemistry.
- Moonens K, et al. 2017. Evolution and Structural Dynamics of Bacterial Glycan Binding Adhesins. Current Opinion in Structural Biology.
- Walz A, et al. 2005. Identification and Characterization of Binding Properties of Helicobacter Pylori by Glycoconjugate Arrays. Glycobiology.
- Orlandi PA, et al. 1992. A Malaria Invasion Receptor, the 175-Kilodalton Erythrocyte Binding Antigen of Plasmodium Falciparum Recognizes the Terminal Neu5Ac (Alpha 2-3) Gal-Sequences of Glycophorin A. Journal of Cell Biology.
- Mehdipour AR, et al. 2021. Dual Nature of Human ACE2 Glycosylation in Binding to SARS-CoV-2 Spike. Proceedings of the National Academy of Sciences.
- Adamczyk B, et al. 2012. Characterization of Fibrinogen Glycosylation and Its Importance for Serum/Plasma N-Glycome Analysis. Journal of Proteome Research.
- Koistinen H, et al. 2003. Differences in Glycosylation and Sperm-Egg Binding Inhibition of Pregnancy-Related Glycodelin. Biology of Reproduction.
- Gough C, et al. 2011. Lipo-Chitooligosaccharide Signaling in Endosymbiotic Plant-Microbe Interactions. Molecular Plant-Microbe Interactions®.
- Moran AP. 2010. Molecular Mimicry of Host Glycosylated Structures by Bacteria. Microbial Glycobiology.
- [21] Scheer, J. M.; Sandoval, W.; Elliott, J. M.; Shao, L.; Luis, E.; Lewin-Koh, S.-C.; Schaefer, G.; Vandlen, R. Reorienting the Fab Domains of Trastuzumab Results in Potent HER2 Activators. PLOS ONE 2012, 7(12), e51817. https://doi.org/10.1371/journal.pone.0051817.
.
[elementor-template id=”4403″][elementor-template id=”4406″]


