Vector Laboratories is closed for the President’s Day on Monday, February 19th. We will be back in the office on Tuesday, February 20th.
We will respond to emails upon our return. Have a wonderful day.
Menu
Vector Laboratories is closed for the President’s Day on Monday, February 19th. We will be back in the office on Tuesday, February 20th.
We will respond to emails upon our return. Have a wonderful day.
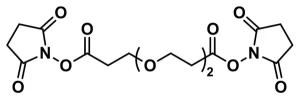
Bis-dPEG®9-NHS ester, product number QBD-10246, is a homobifunctional, amine-reactive, single molecular weight PEG crosslinker with a medium-length (31 atoms, 35.7 Å), discrete PEG (dPEG®) chain length. Each end of the chain terminates as the N-hydroxysuccinimidyl (NHS) ester of propionic acid. Several scientific publications describe the use of QBD-10246.
NHS esters react with free amines such as the ε-amines of lysine with an optimal pH range of 7.0 – 7.5. However, NHS esters can react with free amines with pH as low as 6.0. NHS esters are susceptible to hydrolysis in aqueous media. The hydrolysis rate of the ester increases with the pH.
Thus, we strongly discourage storing Bis-dPEG®9-NHS ester in water or aqueous buffer. Instead, we recommend that customers make new solutions of the product as needed, use them immediately, and discard unused solutions after use. If customers desire to store the product in solution, we recommend the use of a pure, anhydrous, water-miscible solvent such as dimethyl sulfoxide (DMSO), N,N-dimethylacetamide (DMAC), or N,N-dimethylformamide (DMF). DMSO, DMAC, or DMF can be dried chemically or by storing for a minimum of 24 hours over 3 Å molecular sieves. With DMF, use only fresh solvent as the compound decomposes over time to form free amines that will react with the NHS esters.
| Unit Size | 100 mg, 1000 mg |
|---|---|
| Molecular Weight | 708.71; single compound |
| Chemical formula | C₃₀H₄₈N₂O₁₇ |
| CAS | 1008402-79-6 |
| Purity | > 97% |
| Spacers | dPEG® Spacer is 31 atoms and 35.7 Å |
| Shipping | Ambient |
| Typical solubility properties (for additional information contact Customer Support) | Methylene chloride, Acetonitrile, DMAC or DMSO. |
| Storage and handling | -20°C; Always let come to room temperature before opening; be careful to limit exposure to moisture and restore under an inert atmosphere; stock solutions can be prepared with dry solvent and kept for several days (freeze when not in use). dPEG® pegylation compounds are generally hygroscopic and should be treated as such. This will be less noticeable with liquids, but the solids will become tacky and difficult to manipulate, if care is not taken to minimize air exposure. |
Greg T. Hermanson, Bioconjugate Techniques, 3rd Edition, Elsevier, Waltham, MA 02451, 2013, ISBN 978-0-12-382239-0; See Chapter 18, Discrete PEG Reagents, pp. 787-821, for a full overview of the dPEG® products.
Hermanson, G. T. Chapter 5, Homobifunctional Crosslinkers. In Bioconjugate Techniques, 3rd edition; Academic Press: New York, NY, 2013; pp 275–298.
Potent D-peptide inhibitors of HIV-1 entry. Brett D. Welch, Andrew P. VanDemark, Annie Heroux, Christopher P. Hill, and Michael S. Kay. PNAS. 2007, 104 (43) pp 16828-16833. October 23, 2007. DOI: 10.1073/pnas.0708109104.
Dynamic electromagnetophoretic force analysis of a single binding interaction between lectin and mannan polysaccharide on yeast cell surface. Yoshinori Iigunia and Hitoshi Watarai. Analyst. 2010, 135 (6) pp 1426–1432. March 16, 2010. DOI: 10.1039/b924339a.
Orientation-regulated immobilization of Jagged1 on glass substrates for ex vivo proliferation of a bone marrow cell population containing hematopoietic stem cells. Hiroyuki Toda, Masaya Yamamoto, Hiroshi Kohara, Yasuhiko Tabata. Biomaterials. 2011, 32 (29) pp 6920-6928. October 2011. DOI: 10.1016/j.biomaterials.2011.05.093.
Cyclic and dimeric gluten peptide analogues inhibiting DQ2-mediated antigen presentation in celiac disease. Jiang Xia, Elin Bergseng, Burkhard Fleckenstein, Matthew Siegel, Chu-Young Kim, Chaitan Khosla and Ludvig M. Sollid. Bioorganic & Medicinal Chemistry. 2007, 15 (20) pp 6565–6573. October 15, 2007. DOI: 10.1016/j.bmc.2007.07.001.
Phosphoramidate and phosphate prodrugs of (_)-b-D-(2R,4R)-dioxolane-thymine: Synthesis, anti-HIV activity and stability studies. Yuzeng Liang, Janarthanan Narayanasamy, Raymond F. Schinazib and Chung K. Chua. Bioorganic & Medicinal Chemistry. 2006, 14 (7) pp 2178–2189. April 1, 2006. DOI: 10.1016/j.bmc.2005.11.008.
Mapping of Molecular Structure of the Nanoscale Surface in Bio-nanoparticles. Luciana M Herda, Delyan R Hristov, Maria Cristina Lo Giudice, Ester Polo, and Kenneth A Dawson. Journal of the American Chemical Society. 2017. 139(1), 111-114. DOI: 10.1021/jacs.6b12297.
Applicable patents and legal notices are available at legal notices.
Stay in the Loop. Join Our Online Community
Together we breakthroughTM

©Vector Laboratories, Inc. 2024 All Rights Reserved.
How do I Request a Quote?
To request a quote for products: