Description
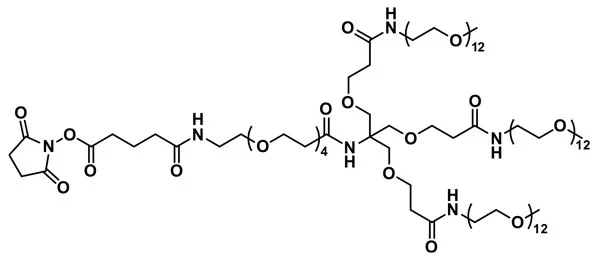
NHS-dPEG®4-(m-dPEG®12)3 ester, product number QBD-10401, is a water-soluble, amine-reactive, single molecular weight, branched PEGylation reagent built around a tris core and used to modify amine-functionalized surfaces and biomolecules. Three equal-length dPEG®12 branches terminate with methyl groups. A short dPEG®4 arm ends with an amine-reactive N-hydroxysuccinimidyl (NHS) ester. From the terminal carbonyl carbon of the dPEG®4 arm to each terminal methyl group on the dPEG®12 branches, the distance is 66 atoms (51.1 Å) long. This hydrophilic branched dPEG® product adds hydrodynamic volume to the conjugates, which reduces immunogenicity and renal clearance. Moreover, this product reduces or eliminates non-specific binding to conjugates and surfaces.
The reaction chemistry for coupling a free amine to an NHS ester is standard in almost all bioconjugation labs worldwide. Numerous scientific publications attest to the benefits of NHS-dPEG®4-(m-dPEG®12)3 ester. These reports describe this product being used to:
Coat nanoparticle surfaces;
Create imaging applications;
Develop Antibody-Drug Conjugates (ADCs);
Modify PK and BD of biomolecular conjugates;
Prevent non-specific binding;
Passivate inorganic surfaces such as metal and diamond; and,
Develop hemerythrin-based blood substitutes.
References
Greg T. Hermanson, Bioconjugate Techniques, 2nd Edition, Elsevier Inc., Burlington, MA 01803, April, 2008 (ISBN-13: 978-0-12-370501-3; ISBN-10: 0-12-370501-0). Specifically see pp. 726-729 in his Chapter 18 on discrete PEG compounds for pegylation applications.
Greg T. Hermanson, Bioconjugate Techniques, 3rd Edition, Elsevier, Waltham, MA 02451, 2013, ISBN 978-0-12-382239-0; See chapter 18, Discrete PEG Reagents, pp.787-821, for a full overview of the dPEG® products.
Towards the Development of Hemerythrin-Based Blood Substitutes. Augustin C. Mot, Alina Roman, Iulia Lupan , Donald M. Kurtz Jr , Radu Silaghi-Dumitrescu. Protein J. 2010, 29 (6) pp 87-393. June 27, 2010. DOI: 10.1007/s10930-010-9264-2.
Rapid Raman Imaging of Stable, Functionalized Nanoshells in Mammalian Cell Cultures. Yiming Huang, Vimal P. Swarup, Sandra Whaley Bishnoi. Nano Letters. 2009, 9 (8) pp 2914-2920. May 23, 2009. DOI: 10.1021/nl901234x.
Differential patterning of neuronal, glial and neural progenitor cells on phosphorus-doped and UV irradiated diamond-like carbon. Edward M. Regan, James B. Uney, Andrew D. Dick, Yiwei Zhang, Jose Nunez-Yanez, Joseph P. McGeehan, Frederik Claeyssens, Stephen Kelly. Biomaterials. 2010, 31 (2) pp 207-215. October 14, 2009. DOI: 10.1016/j.biomaterials.2009.09.042.
Different radiolabelling methods alter the pharmacokinetic and biodistribution properties of Plasminogen Activator Inhibitor Type 2 (PAI-2) forms. Marie Ranson, Paula Berghofer, Kara L. Vine, Ivan Greguric, Rachael Shepherd, Andrew Katsifis. Nuclear Medicine and Biology. 2012, 39 (6) pp 833-839. January 17, 2012. DOI: 10.1016/j.nucmedbio.2012.01.006.
Circulation and long-term fate of functionalized, biocompatible single-walled carbon nanotubes in mice probed by Raman spectroscopy. Zhuang Liu, Corrine Davis, Weibo Cai, Lina He, Xiaoyuan Chen, and Hongjie Dai. PNAS. 2008, 105 (5) pp 1410-1415. February 5, 2008. DOI: 10.1073_pnas.0707654105.
Synthesis and Preliminary Biological Evaluation of High-Drug-Load Paclitaxel-Antibody Conjugates for Tumor-Targeted Chemotherapy. Sherly Quiles, Kevin P. Raisch, Leisa L. Sanford, James A. Bonner, and Ahmad Safavy. Journal of Medicinal Chemistry. 2009, 53 (2) pp 586-594. December 3, 2009. DOI: 10.1021/jm900899g.
TIRF microscopy as a screening method for non-specific binding on surfaces. Christy Charlton, Vladimir Gubala, Ram Prasad Gandhiraman, Julie Wiechecki, Nam Cao Hoai Le, Conor Coyle, Stephen Daniels, Brian D. MacCraith, David E. Williams. Journal of Colloid and Interface Science. 2011, 354 (1) pp 405-409. February 1, 2011. DOI: 10.1016/j.jcis.2010.10.029.
Influence of PEGylation with linear and branched PEG chains on the adsorption of glucagon to hydrophobic surfaces. Charlotte Pinholt , Jens Thostrup Bukrinsky, Susanne Hostrup, Sven Frokjaer, Willem Norde, Lene Jorgensen. European Journal of Pharmaceutics and Biopharmaceutics. 2010, 77 (1) pp 139–147. November 3, 2010. DOI: 10.1016/j.ejpb.2010.11.001.
The influence of polyethylence glycol structure on the conjugation of recombinant human interferon a2b overproduced using synthetic gene in E. coli. Heni Rachmawati, Prima L. Febrina, Ratih A, Ningrum, Debbie S. Retnoningrum. International Journal of Research in Pharmaceutical Sciences. 2012, 3(2) pp 228-233. March 20, 2012. ISSN: 0975-7538.
Monitoring Protein PEGylation with Ion Exchange Chromatography. Peter Yu, Deanna Hurum, Leo Wang, Terry Zhang,and Jeffrey Rohrer. ThermoFisher Scientific Poster Note. 2013, PN70510_E01/3s.
New Assay for Quantification of PEGylated Proteins During in Vitro Permeability Studies. Spela Jalen, Vanja Smilovic, Katarina Fidler, Barbara Podobnik, Maja Marusic, Radovan Komel, Vladka Gaberc-Porekar and Simona Jevsevar. Acta Chimica Slovenica. 2014, 61 (3) pp 615-622. January 15, 2014.
Nanometer size silicon particles for hyperpolarized MRI. Grzegorz Kwiatkowski, Fabian Jahnig, Jonas Steinhauser, Patrick Wespi, Matthias Ernst, and Sebastian Kozerke. Scientific Reports. 2017, 7 (7946) pp 1-9. August 11, 2017. DOI: 10.1038/s41598-017-08709-0.
Aptamer-Modified Nanohydrogel Microarrays for Bioselective Cancer Cell Immobilization. Zan Lamberger, Hendrik Bargel, Martin Humenik. Advanced Functional Materials, Volume 32, Issue 45 (2022). 08/28/22. https://doi.org/10.1002/adfm.202207270